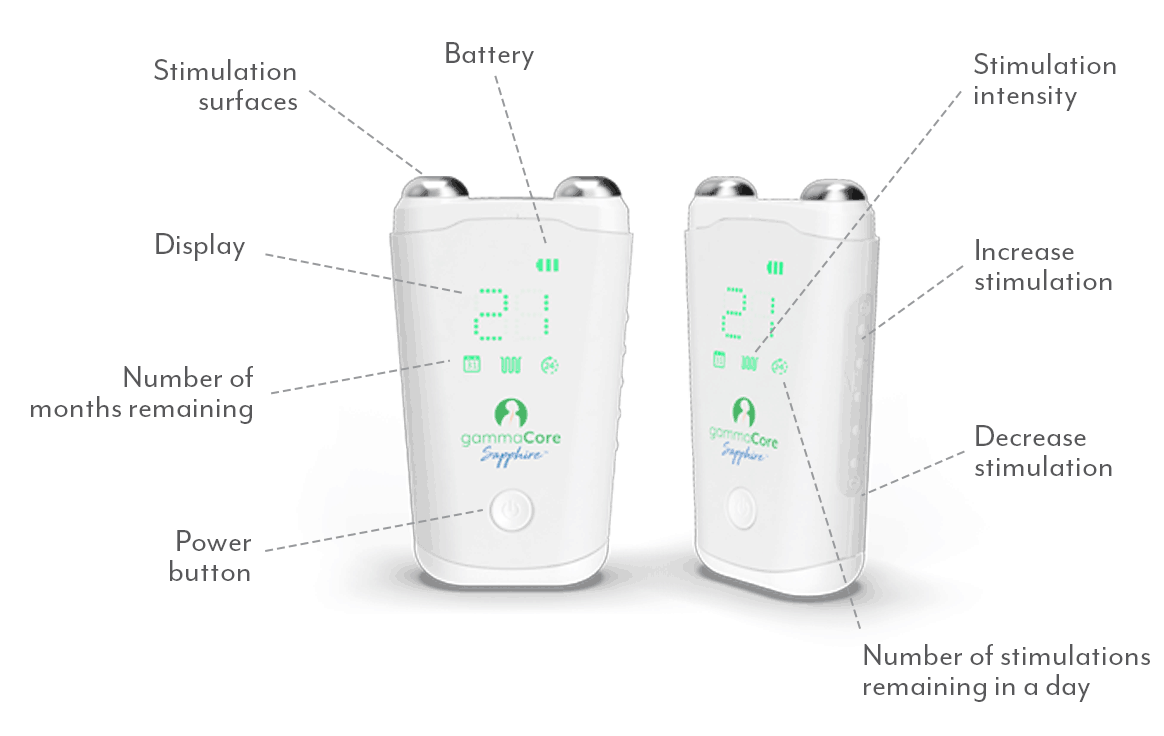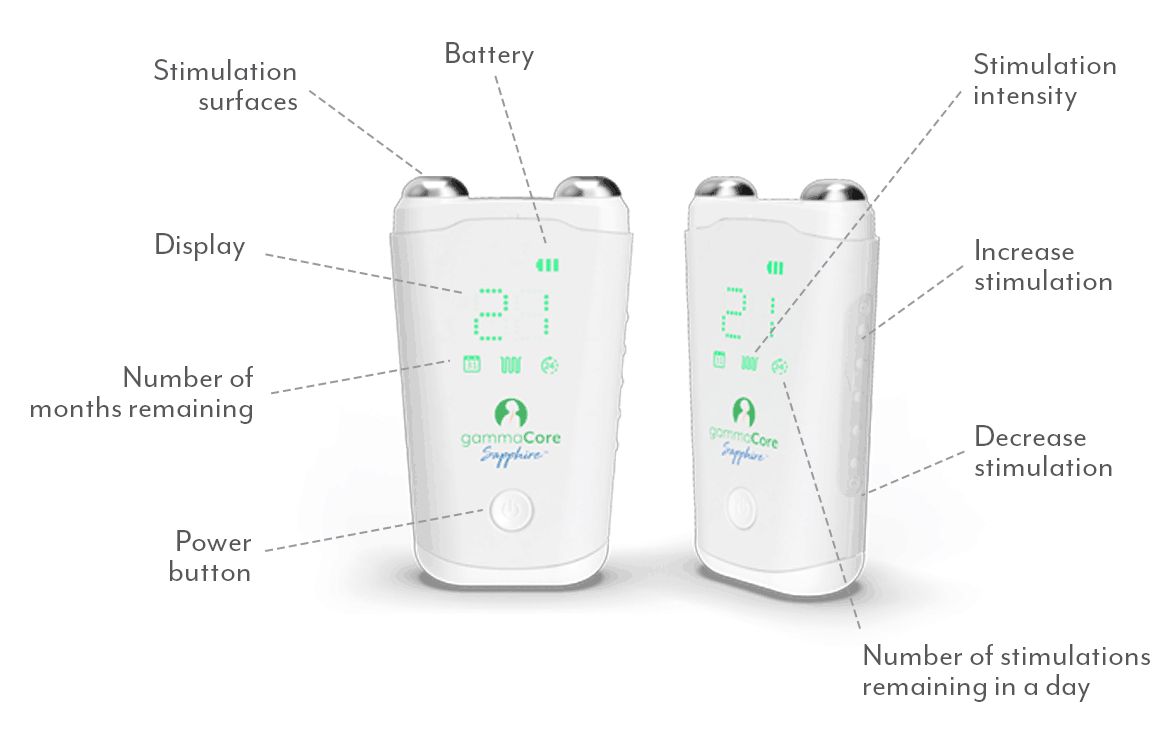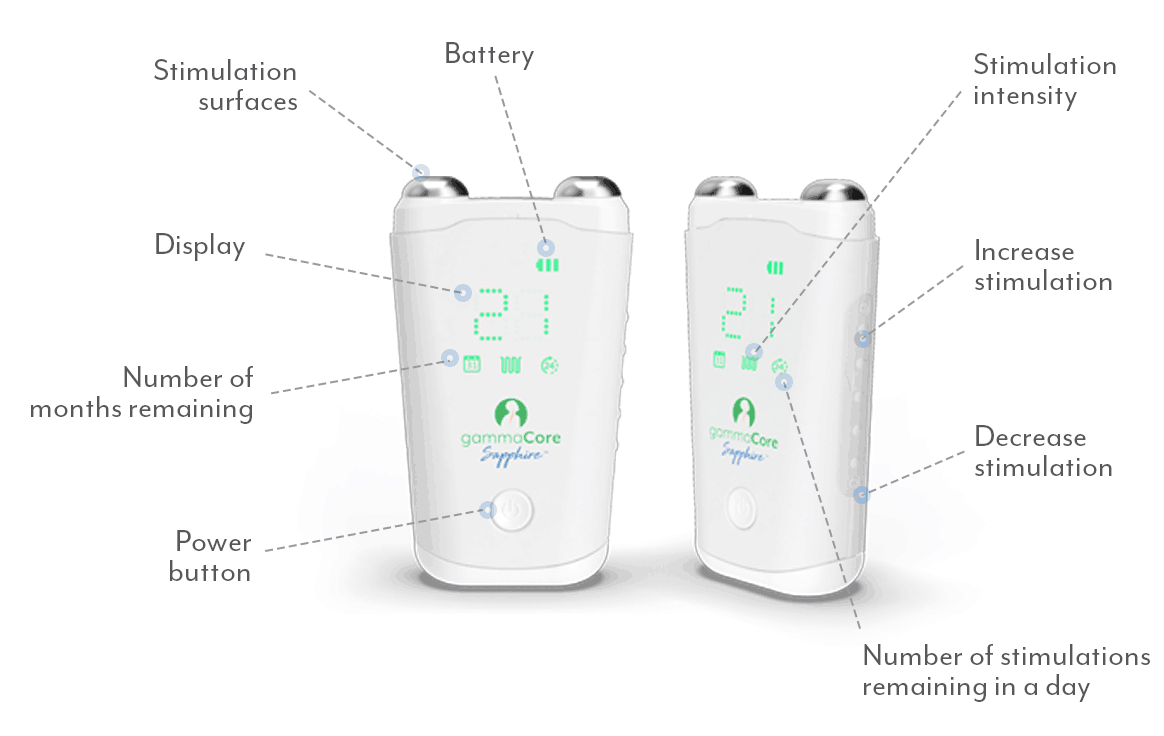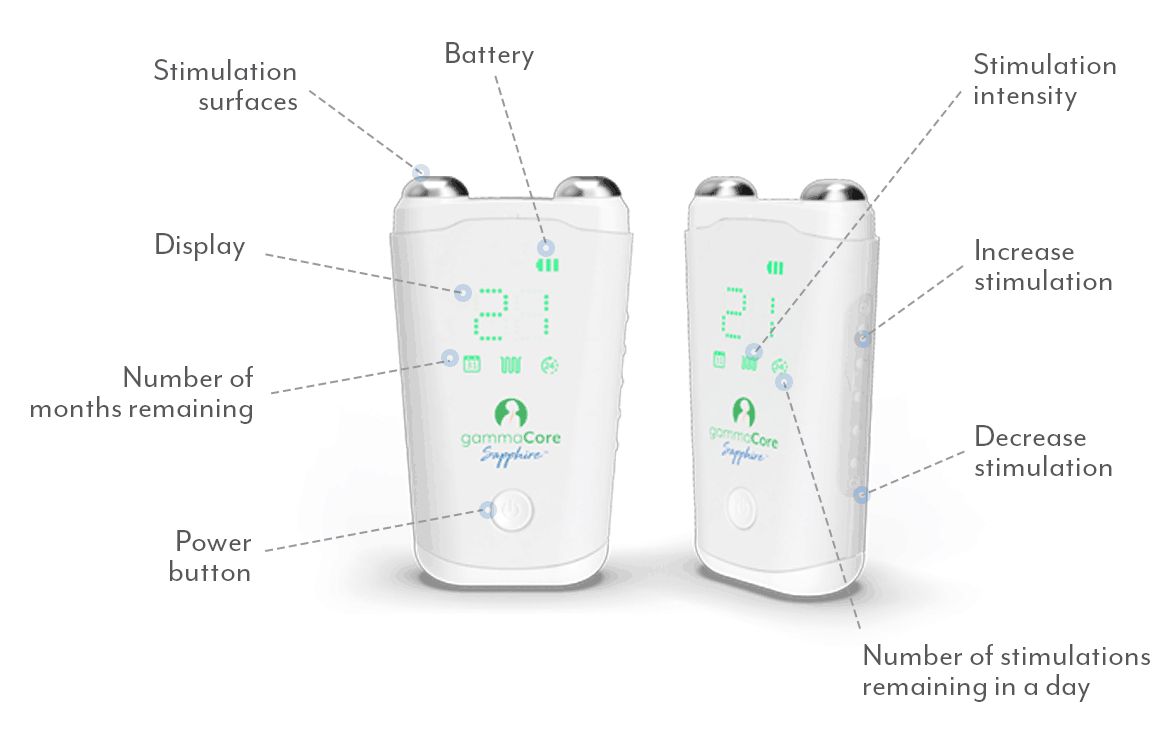
How Does gammaCore Work?
Unlike an implanted vagus nerve stimulator or other migraine and cluster headache treatments, the gammaCoreTM non-invasive vagus nerve stimulator, otherwise known as nVNS, activates the vagus nerve with gentle electrical stimulation through the skin via the neck. Helping to prevent and relieve headache pain without pills, injections and drug-like side effects.
What is the Vagus Nerve?
The vagus nerve is the longest cranial nerve in the body, with branching nerves that go to the heart, lungs, stomach, and many other organs. It primarily serves as a sensory nerve, responsible for reporting information to the brain and transmitting instructions back to the body.
Think of the vagus nerve as the nervous systems “super-highway”, sending important communication between your brain and many parts of the body and back. It facilitates and communicates with many different systems, operating and influencing things like pain regulation, anxiety, fatigue, heart rate, digestive track activity, and lung function.
How does gammaCore stimulate the Vagus Nerve?
As the longest cranial nerve in your body, part of the vagus nerve passes through the neck as it travels between the chest, abdomen, and lower part of the brain. During treatments, gammaCore can be placed on either side of the neck where the vagus nerve is located and sends proprietary gentle electrical energy non-invasively through the skin. The electrical energy stimulates the vagus nerve and begins sending messages to the brain.
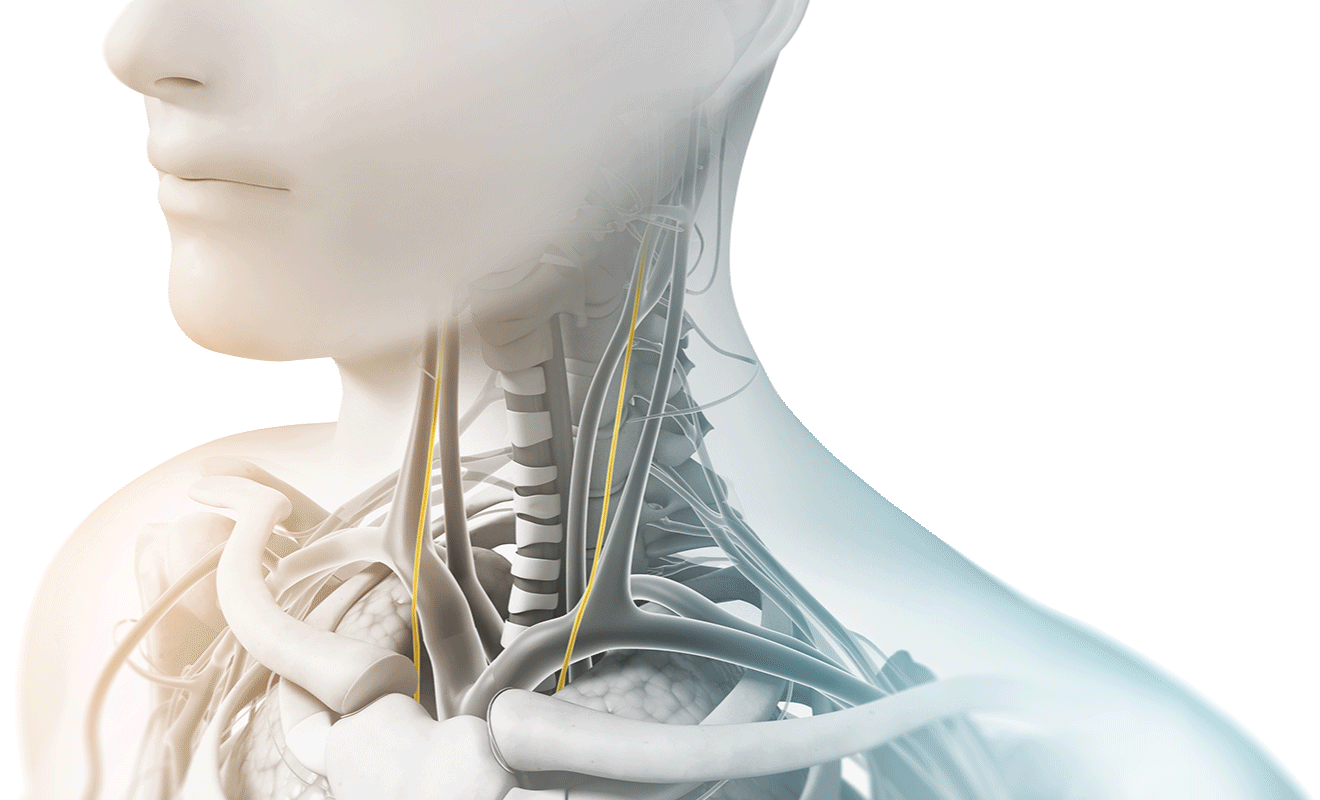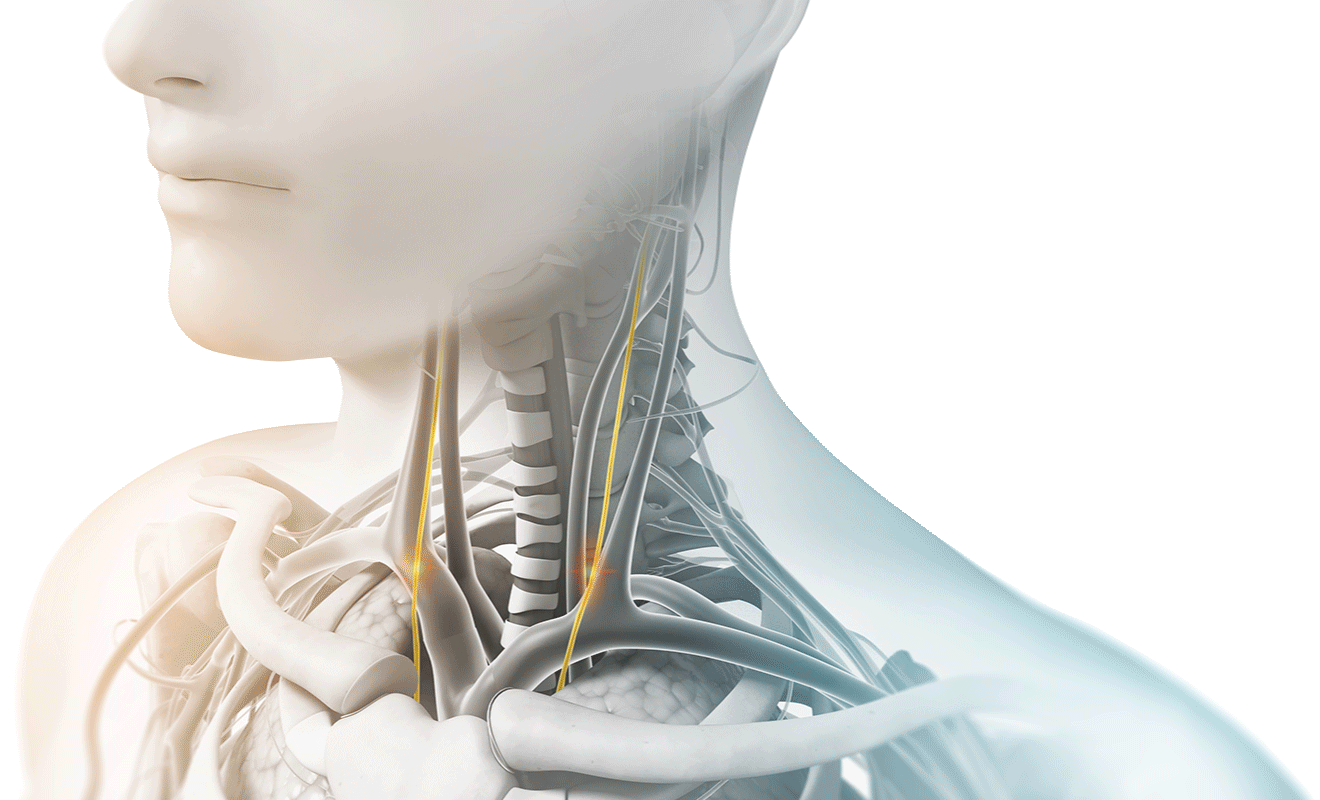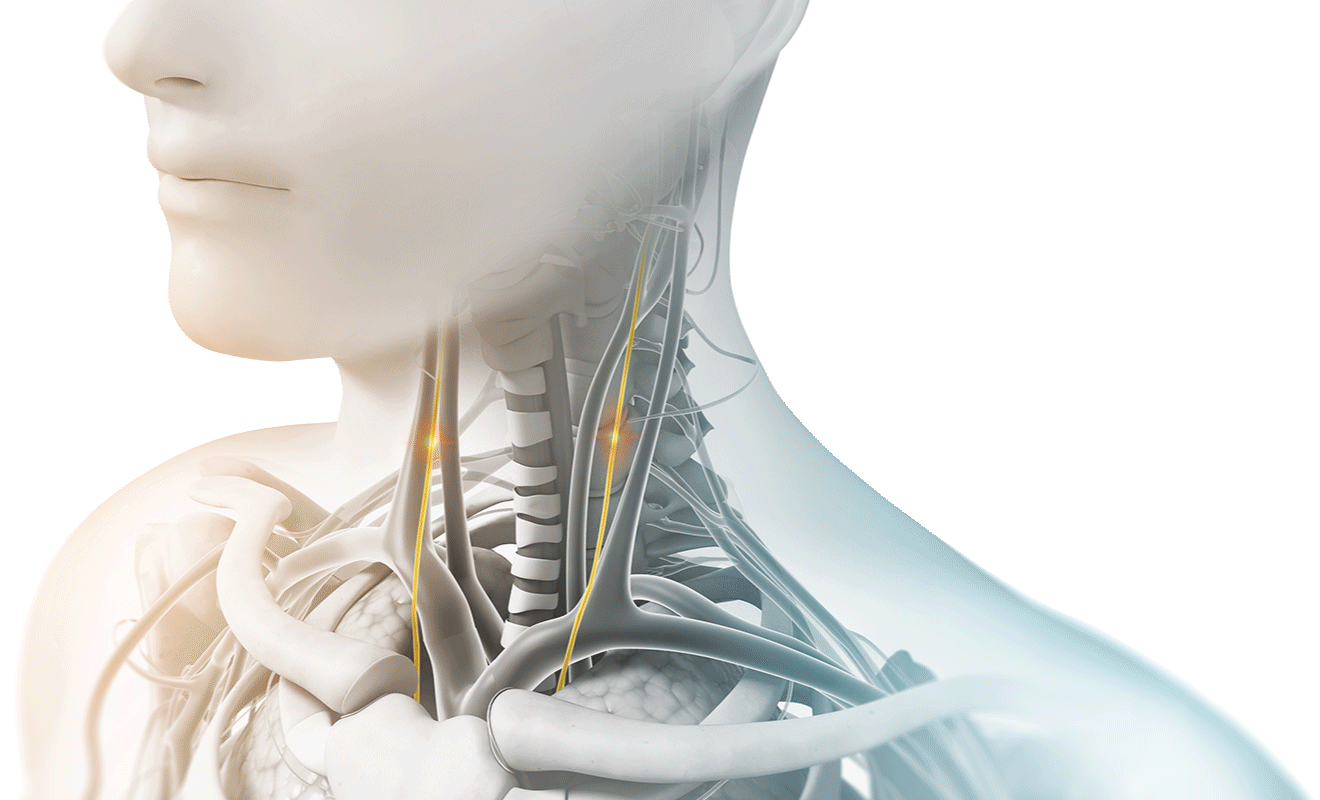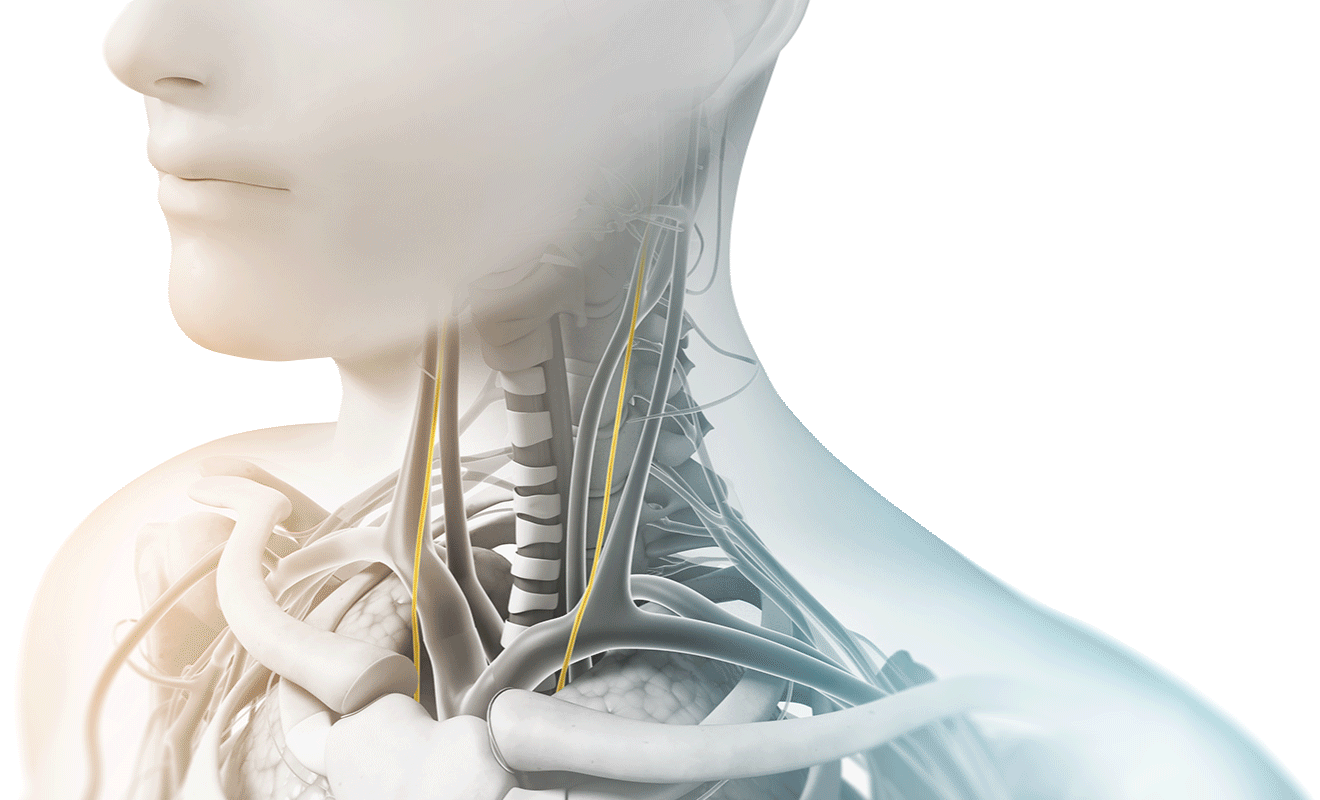
How does Vagus Nerve Stimulation help those suffering from headache pain?
The vagus nerve plays an important role in regulating pain. When stimulating the vagus nerve, pain signals causing the attacks can be blocked, helping provide fast relief and preventing future attacks before they happen.
Experience more headache-free days without worrying about drug overuse or inconveniences associated with injectable, inhalers, and medications.

Backed by Research
Vagus nerve stimulation has been widely researched and studied. The gammaCore device itself has 7 different FDA clearances with robust scientific & clinical data to support its safe and effective use, including >7 randomized controlled clinical trials, >30 mechanisms of action peer-reviewed papers, and >40 peer-reviewed clinical papers.
Evidence of activation of vagal afferents by non-invasive vagus nerve stimulation: An electrophysiological study in healthy volunteers
Romain Nonis, Kevin D'Ostilio, Jean Schoenen, Delphine Magis; Nov 2017“Cervical nVNS can activate vagal afferent fibers, as evidenced by the recording of far-field vSEPs similar to those seen with iVNS and non-invasive auricular stimulation.”
Noninvasive vagus nerve stimulation as acute therapy for migraine: The randomized PRESTO study
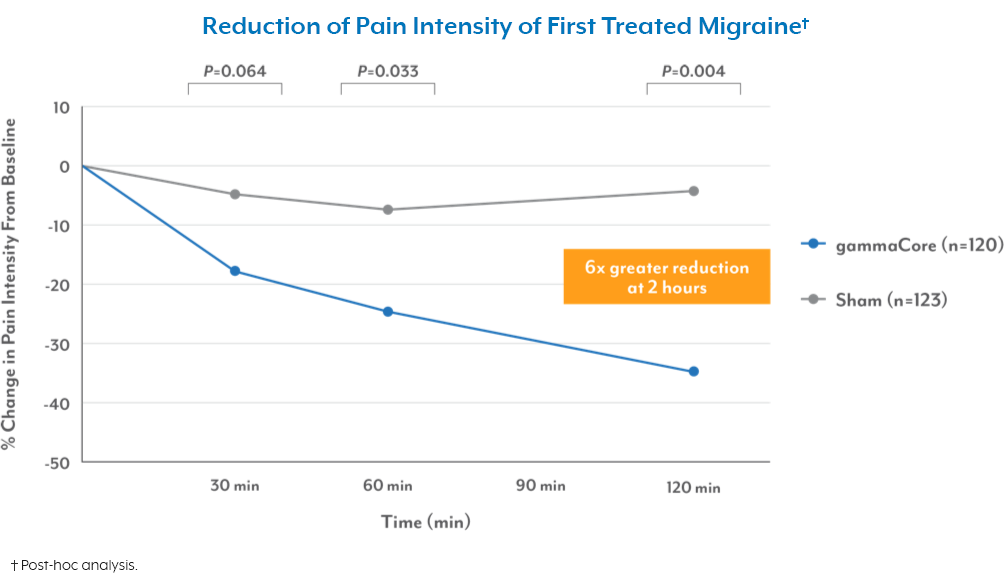
Cristina Tassorelli, Licia Grazzi, Marina de Tommaso, Giulia Pierangeli, Paolo Martelletti, Innocenzo Rainero, Stefanie Dorlas, Pierangelo Geppetti, Anna Ambrosini, Paola Sarchielli, Eric Liebler, Piero Barbanti, PRESTO Study Group; July 2018“This randomized sham-controlled trial supports the abortive efficacy of nVNS as early as 30 minutes and up to 60 minutes after an attack. Findings also suggest effective pain relief, tolerability, and practicality of nVNS for the acute treatment of episodic migraine.”
Vagus nerve stimulation for prevention of migraine: The multicenter, randomized, double-blind, sham-controlled PREMIUM II trial
Umer Najib, Timothy Smith, Nada Hindiyeh, Joel Saper, Barbara Nye, Sait Ashina, Candace K McClure, Michael J Marmura, Serena Chase, Eric Liebler, Richard B Lipton; June 2022“These results suggest clinical utility of non-invasive vagus nerve stimulation for migraine prevention, particularly for patients who have migraine with aura, and reinforce the well-established safety and tolerability profile of this therapy.”