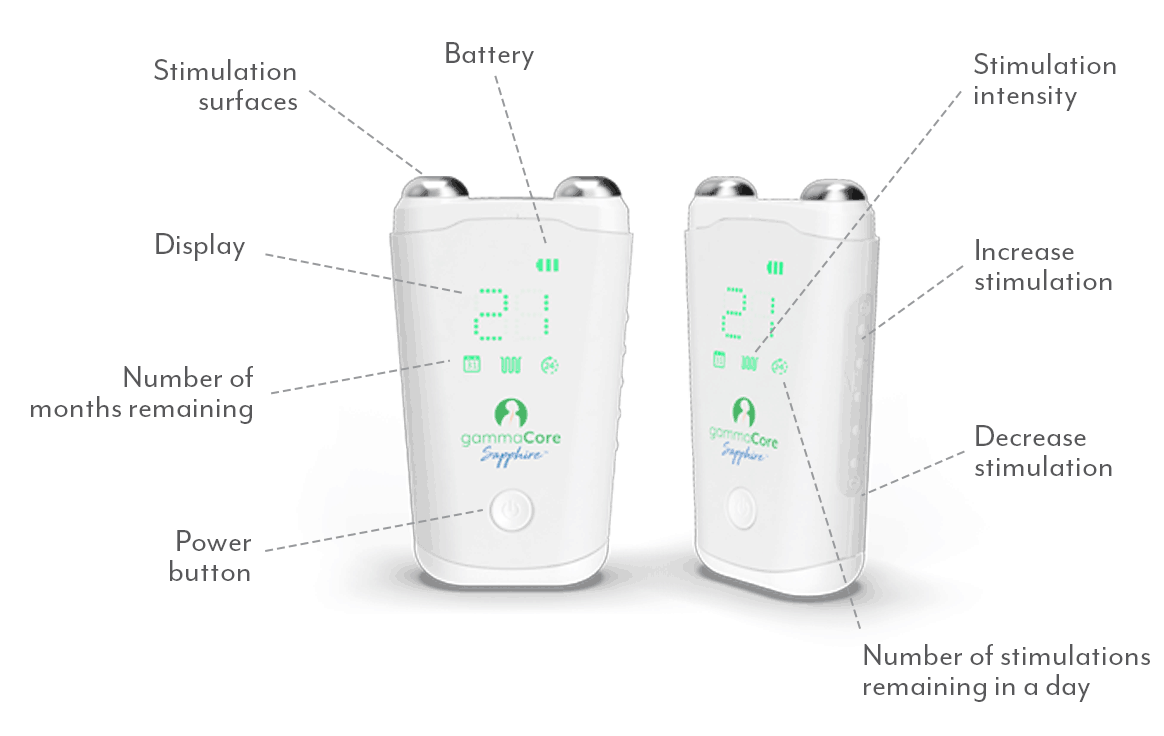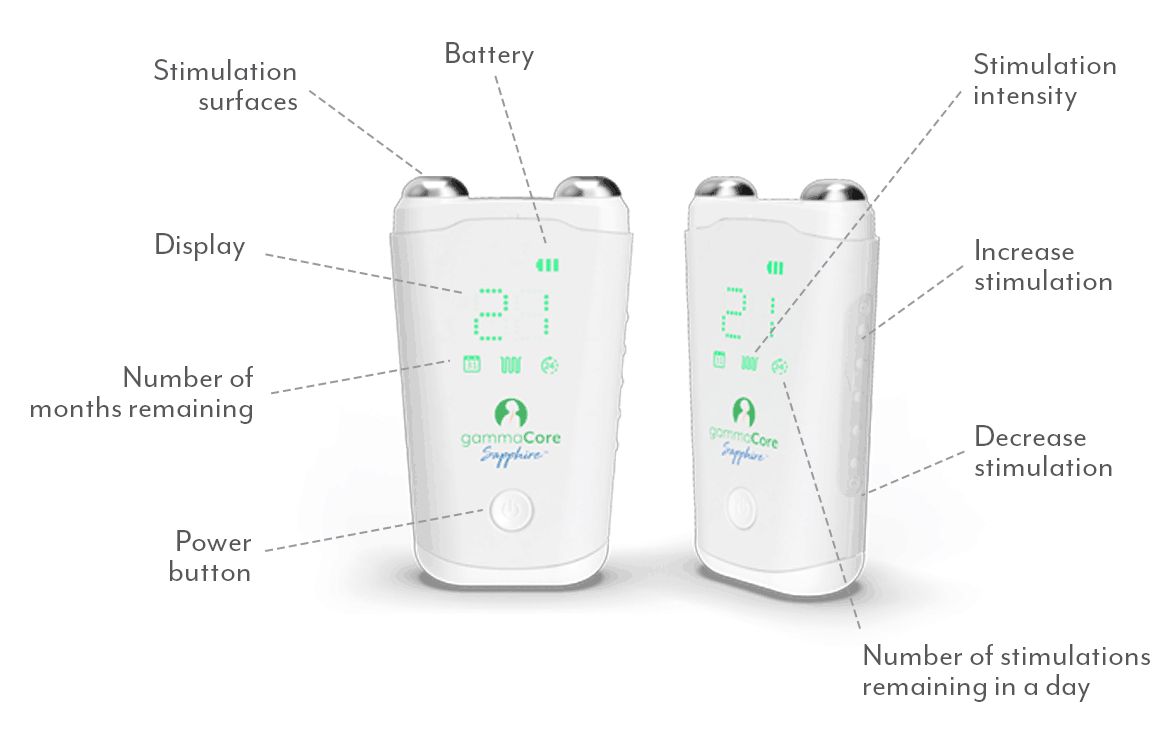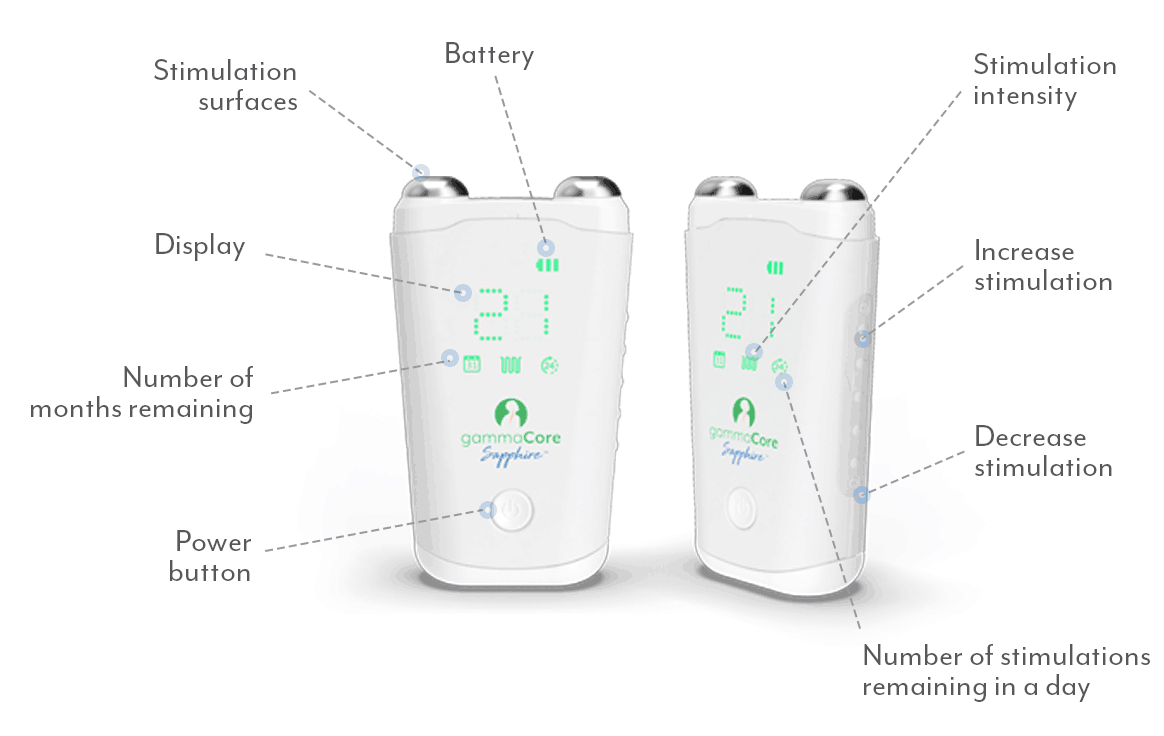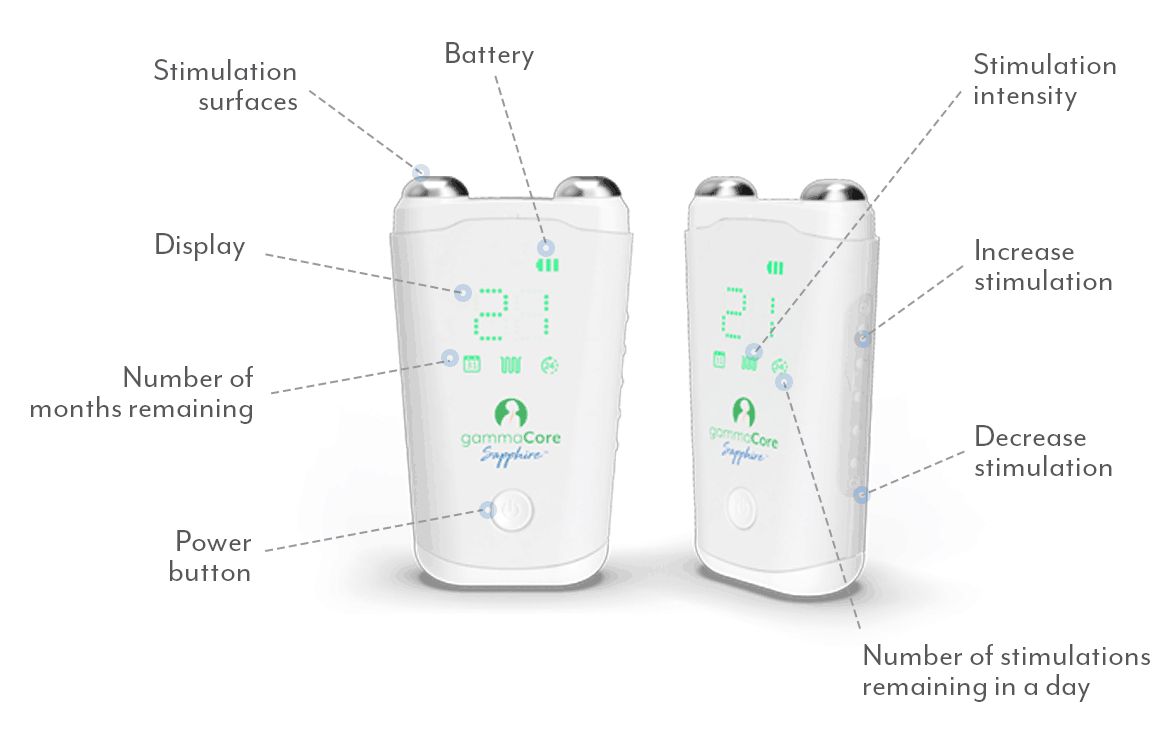
Important Safety and Performance Information
electroCore, Inc. provides easy access to gammaCore™ non-invasive vagus nerve stimulation (nVNS) product information, including online and printed materials. Below, you’ll find Important Safety and Performance Information for select products. We recommend downloading, printing, and keeping these documents available for reference when using gammaCore nVNS.
To download and view Important Safety and Performance Information, click on the links below:
If you experience any difficulty accessing these documents or would like to request a printed copy at no cost, please contact our dedicated Customer Experience team at CustomerService@electroCore.com or 888-903-2673.