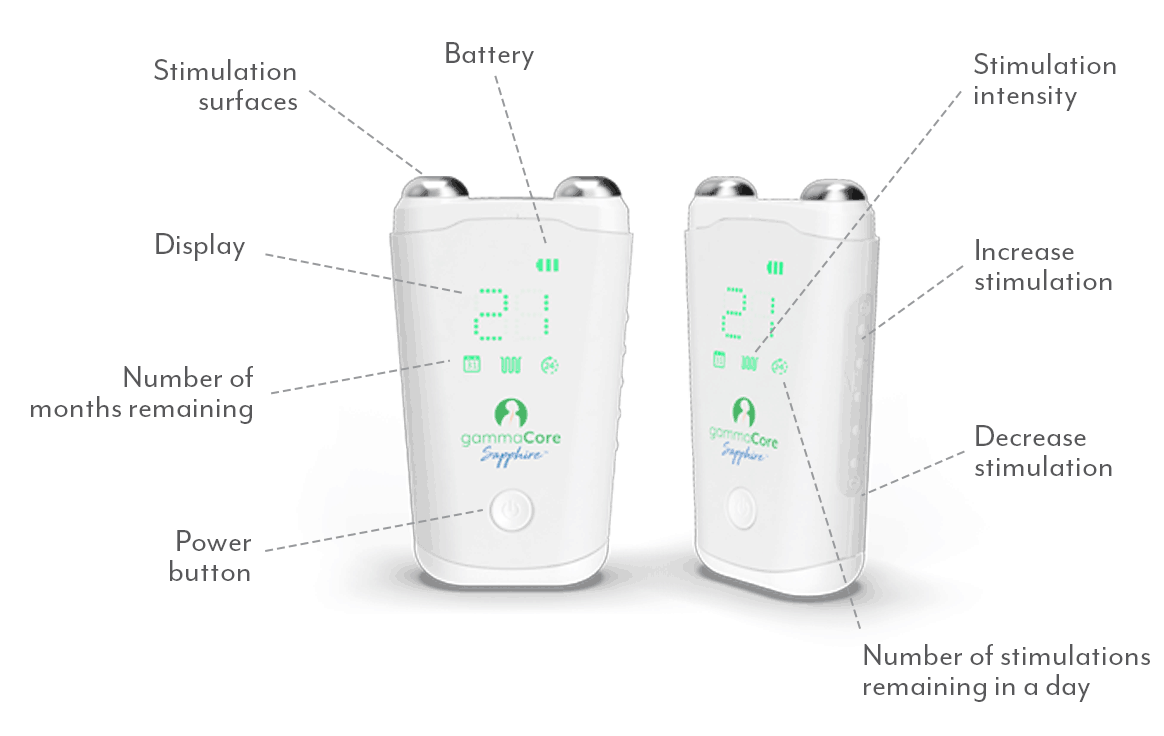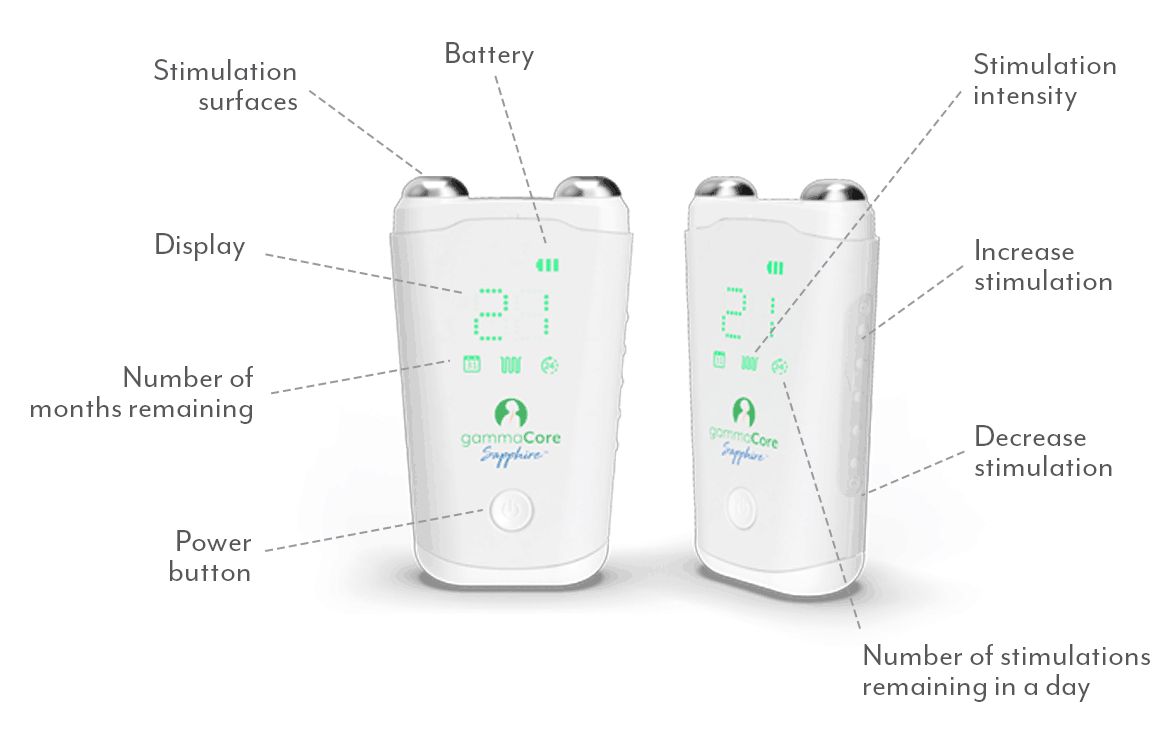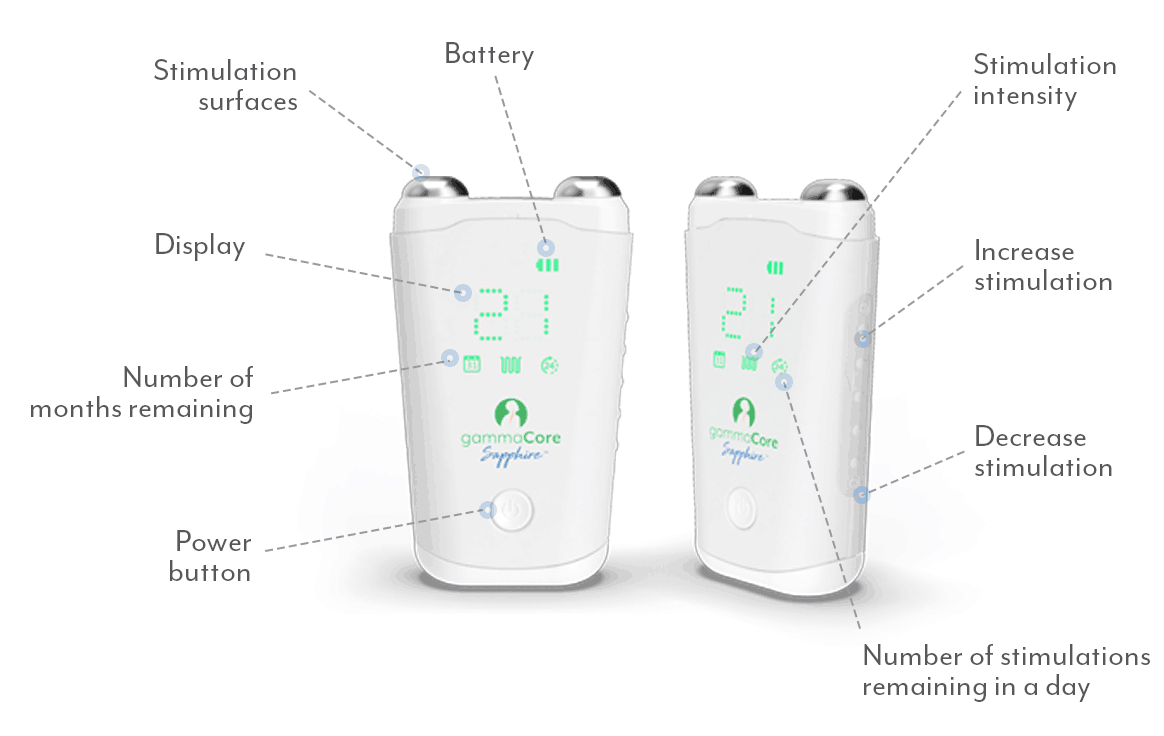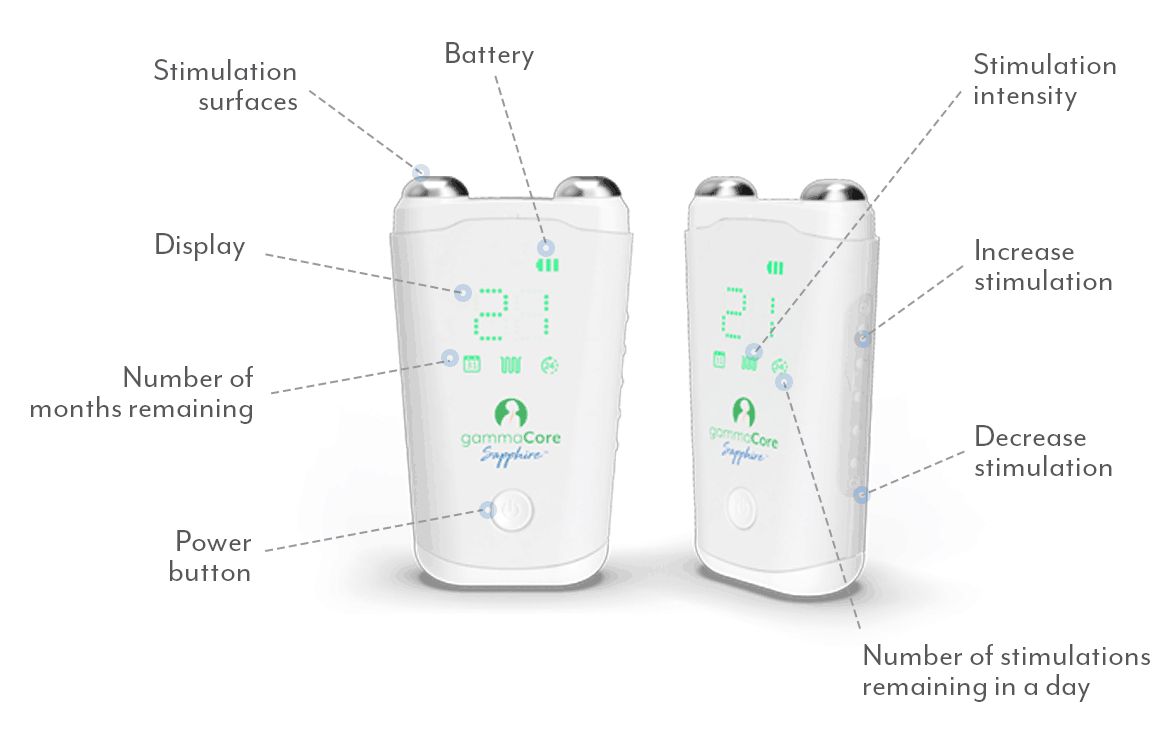
One Device. Many Uses.

gammaCore SapphireTM is the first and only FDA cleared non-invasive device to treat and prevent multiple types of headache pain via the vagus nerve. It’s small, handheld and portable for quick and easy treatments whenever and wherever needed.
For acute use:
Treat right away and with a few 2-minute treatments, you could see fast acting, long-lasting pain relief.
To prevent future headache pain:
Simply treat 2 times a day (morning and night) for more headache-free days, so you can live life with less disruption.
Safe, Effective and Easy to Use Treatment For

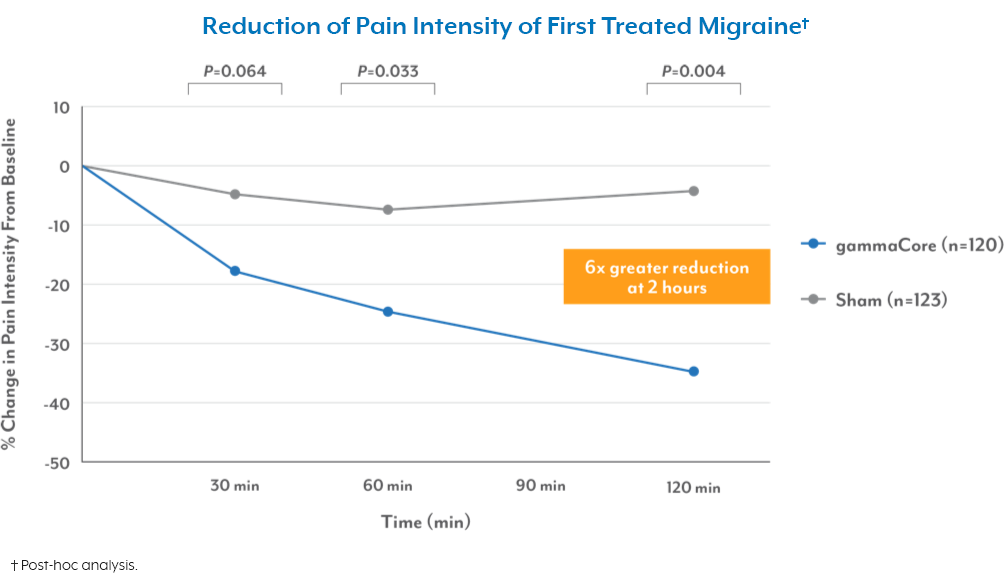
Migraine
Clinically proven to treat and prevent migraines in users ages 12 and up. Help reduce migraines days without additional drugs.
Learn More
Cluster Headache
The only clinically proven device to treat and prevent cluster headache attacks, while providing fast, reliable relief for those suffering with cluster headache.
Learn More
Hemicrania Continua (HC) & Paroxysmal Hemicrania (PH)
1st and ONLY device for HC and PH treatment, proven to reduce pain severity or headache frequency.
Learn MoreHow It Works
Unlike traditional vagus nerve stimulation, gammaCore non-invasive vagus nerve stimulator (nVNS) activates the vagus nerve with gentle electrical stimulation through the skin to prevent and relieve headache pain, without the need for surgery.
Why stimulate the vagus nerve? Think of the vagus nerve as the “superhighway” of the nervous system. This important highway sends communications between your brain and many parts of the body.
Stimulating the vagus nerve blocks the pain signals causing the attacks, to help provide fast relief and prevent future attacks.
See How gammaCore Is Changing Lives

Marie
gammaCore User
“gammaCore was awesome, and gave me immediate pain relief. gammaCore has been really convenient for me.”
See Her Story
Tracey
gammaCore User
“I started feeling like myself again. I am not afraid migraine is going to control my life. For me gammaCore works.”
See Her Story
Jess
US Navy Veteran - gammaCore User
“With gammaCore, I am able to start doing things and I feel like a human being again.”
See His Story
Dr. Sara Sacco
gammaCore Prescriber
“It’s so easy and convenient to use. I have really been so glad to have another alternative to treat my patients.”
See Her Story
Morgan
gammaCore User
“I've been a super responder to gammaCore. I was a non-responder to everything else. With daily use of gammaCore as a preventive therapy I rarely need rescue meds now.” *

gammaCore User
"I have cluster headaches every day and I know I can use gammaCore easily and discretely several times a day and be included in activities with family and friends. No longer am I experiencing daily trauma and having my loved ones watching it." *
*Individual results may vary
Featured In


“Digital Health’s
Big Secret”

“gammaCore can be used as often as you need, isn’t invasive, and doesn’t come with the side effects of other treatments”

“Cutting-Edge Treatments
for Migraines”

“The Best Advances to Stop Chronic Headaches”
Ready to Get Started? Here's How

Step 1
Download the prescription form.

Step 2
Have your healthcare provider complete the form, then email the completed form to orders@electrocore.com or fax 1-855-647-1320.

Step 3
Once received, we will reach out within 1-3 days to confirm details.

Your gammaCore expenses may be eligible for reimbursement through your Health Savings Account (HSA) or Flexible Saving Account (FSA). Ask us for more details!